Best IHC Staining Practices
Fixation
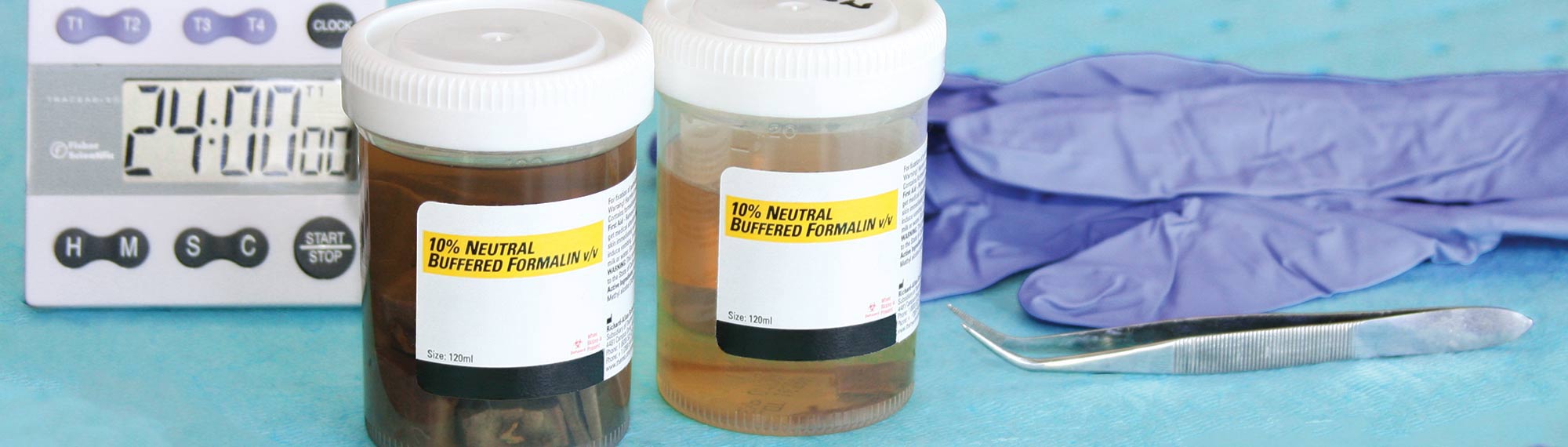
Time: 12–48 hours; tissue must be fixed immediately upon removal from the body
Underfixation or delayed fixation can result in:- Weak or No staining proteins are not preserved and thus subject to denaturation. If the target protein is denatured or distorted it will become unrecognizable by the primary antibody and there will be no antibody-epitope binding.
- Background staining autolysis, or cellular self-digestion, can occur when tissue is removed from the body and not preserved properly or quickly enough. Primary antibody and chromogen can bind non-specifically to the resulting necrotic cells and cellular components.
Overfixation can result in:
Tech Tip: In most cases, antigenicity loss caused by overfixation can be restored by using a more intense pretreatment protocol.
Temperature: Room temperature
Background staining while the addition of heat may shorten the standard fixation time, it will also increase the rate of autolysis.
Tech Tip: Heat acts as a catalyst to speed up a chemical reaction. In the case of fixation, this involves the formation of cross-links between proteins.
Type: 10% Neutral Buffered Formalin (NBF)
Weak or No staining use of other fixatives like alcohols, Mercurials, Bouin’s, Schaffer’s, etc. may result in a lack of adequate protein preservation. These fixatives may also damage or alter target epitopes. If the target antigen/epitope is damaged or denatured, the antibody cannot bind.
Tech Tip: 10% NBF is the optimal fixative for preserving proteins; proteins are your antibody’s target.
| Target to Fix | Preferred Fixative |
|---|---|
| Proteins | 10% NBF |
| Enzymes | Frozen sections |
| Lipids | Frozen sections, Glutaraldehyde |
| Nucleic Acids | Alcohol |
| Biogenic Amines | Bouin’s, 10% NBF |
| Glycogen | Alcohol |
Volume: 15–20x greater than the volume of the tissue specimen
Weak or Variable staining insufficient volume will result in limited fixative penetration; the edges of the tissue will be well preserved, but the center of the tissue will not. Staining is likely to occur optimally around the well-preserved edges, but not toward the under-preserved center.
Tech Tip: A tissue specimen must be fully immersed in fixative to allow for adequate and even tissue preservation.
Microtomy

Embedding: Paraffin ensures cellular morphology is adequately supported; protocols vary
Tech Tip: Proper fixation is critical for the successful creation of model, morphologically sound tissue sections.
Under-filled cassette:
- Unstable microtome clamping causes uneven sections (thick/thin) that can result in interpretation issues for the pathologist.
- Misalignment of the block face causes uneven sections (thick/thin) that can result in interpretation issues for the pathologist.
- Depending on tissue type, inappropriate orientation can cause sectioning difficulties and interpretation issues.
Baking: 2–4 hours at 60°C
No staining If the slides are not baked adequately, water trapped underneath the tissue and/or poor tissue adhesion to the glass slide can result in damaged or lost tissue during heated pretreatment.
Tech Tip: Fatty tissues like breast do not adhere to microscope slides as well or as strongly as other tissues; baking overnight at 60ºC is recommended to prevent tissue loss during pretreatment.
Sectioning:
- Ideal tissue thickness: 3–4µm
- Blade: sharp, clean, properly angled (~5°)
- Water bath: ideal temperature: 35–40°C (no additives)
- Positively charged slides
Tech Tip: To ease sectioning, it is best to place the tissue block on a cold, slightly wet surface. It is important not to freeze the block however, this can create cracks and may increase separation from the paraffin wax.
Poor quality sections can cause a variety of staining and interpretation issues:
- Folds, compression, and knife lines clearance angle too great and/or dull, nicked blade
- Chatter and cracks insufficient clearance angle and/or inappropriate cutting rate
- Disruption, folds, and over-expansion of tissue water bath too hot
Weak or No staining slide adhesive in water bath interferes with IHC reagents.
No staining positively charged glass slides help your negatively charged tissue adhere. Not using these slides can result in tissue loss during heated pretreatment.